Despite the name, the substance is essential to life.
Nitrogen
In this article, we'll look at the characteristics of nitrogen in chemistry, learn what oxidation states nitrogen can have, and talk about the most important compounds that include this chemical element.
Nitrogen (N2) is the first member of group V of the main subgroup and period 2 of the periodic table of elements by D. Mendeleev. If we consider location of nitrogen in long-period Mendeleev table, it occupies the leading place in group 15. For the representatives of this group, the name has been proposed pniktogenes (from the Greek root πνῑ́γω – suffocating, bad smelling). This clearly refers to the hydrogen compounds of representatives of this group.
The electronic structure of nitrogen
Consider the atomic structure and electronic configuration of nitrogen, and then draw some conclusions.
The atomic or ordinal number of nitrogen is 7, which corresponds to the number of electrons and protons in the nucleus. The molar mass is 14.00728 g/mol, and the number of neutrons in an atom of this isotope is seven.
Now let's move on to the electronic structure. In the ground state, the electronic formula for nitrogen is: 1s 2 2s 2 2p 3 , abbreviated as [He]2s 2 2p 3 . There are 5 valence electrons on the outer energy level, including 3 unpaired p-electrons.
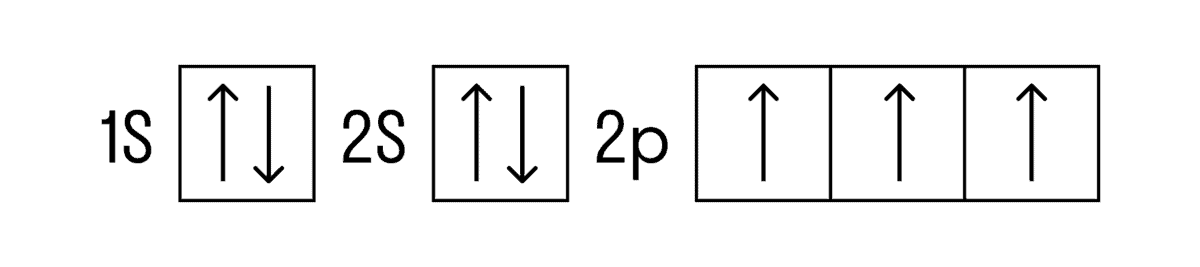
Based on this configuration, nitrogen can form only three bonds by the exchange mechanism and one more by the donor-acceptor mechanism. This is due to the fact that there are no more vacant orbitals of nitrogen on the second sublevel where electrons from the 2s-level can evaporate. This results in the maximum valence of nitrogen IV.
Nitrogen is characterized by the entire spectrum of possible oxidation degrees from -3 to +5.
Let's take a look at the scale, which reflects nitrogen compounds in different substances.

Get tips, articles, videos and training checklists in your inbox
![]()

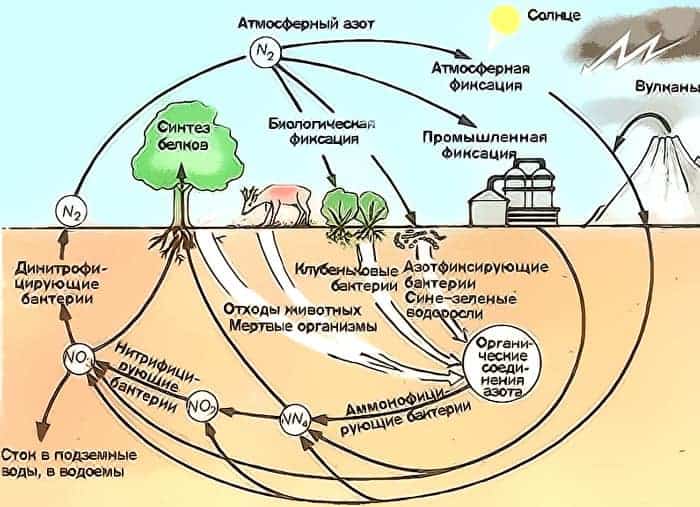
Nitrogen in Nature
It is one of the most abundant chemical elements on the planet. Large amounts of it are present in the atmosphere, in parts of plants and living organisms – in the form of nitrates, oxides, nucleic acids, amino acids, proteins, and more.
As a fossil it is poorly distributed, most of all as saltpeter and ammonium chloride.
For science and technology, of particular interest is the highest hydroxide of nitrogen, more precisely – nitric acid. Its smell, specific ammonia, is known to everyone.
Prevalence
It is estimated that the Earth's atmosphere alone contains about 3.8 x 10 to the 15th power of tons of nitrogen. Up to another 1.5 x 10 to the 15th power of tons is found in the surface layers of our planet, and in the mantle – about 1.3 x 10 to the 16th power of tons.
The latter is calculated taking into account the actual data on the amount and composition of lava erupting from volcanoes: it contains a huge amount of nitrogen.
Space is also rich in it: it is found on several planets of the solar system and their satellites, in gas clusters. It is many billions of tons.
Isotopes
The chemistry of nitrogen is diverse, with a number of known oxides. All of them are interesting with a wide range of N oxidation degrees and form "their" acids and salts:
- N2O (the oxidation degree of the nitrogen molecule is +1, the lowest possible), a sweet-smelling gas used for narcosis: it forms hypo-nitric acid and hyponitrite (the name of the derivative salt);
- NO (+2), is poisonous at high concentrations: nitroxyl acid and nitroxylate;
- N2O3 (+3), exists in pure form only at negative temperatures: nitrite;
- NO2 or N2O4 (+4), colorless under normal conditions and brown if the accompanying heating is introduced;
- N2O5 (+5), volatile crystals that are hygroscopic.
Obtaining
The atom and molecule of pure nitrogen are optimally isolated under laboratory conditions. To do this, the following chemical reactions are used:
- The reduction of sodium nitrite with ammonium chloride yields water and nitrogen;
- combustion of ammonia;
- Thermal decomposition of some alkali metal compounds;
- ammonia reduction of oxidized copper, and a number of others.
Under industrial conditions, membrane, cryogenic and adsorption methods are used. Each of them requires equipment and special conditions of performance.
Further processing produces the highest nitrogen hydroxide, nitric acid (HNO3), which can react with a wide range of substances.
Decomposition of ammonium nitrite
The exothermic decomposition reaction of nitrite needs cooling – the heat release is about 330 kJ. The initial period is interesting – short-term heating of the product is necessary to activate the reaction.
![]()
Ammonium sulfate is added in small quantities to a vessel with ammonium nitrite. As a result, nitrogen and water are released, contaminated with small amounts of oxygen and ammonia.
It is possible to get rid of them by sequentially running the reaction products through sulfuric acid, iron sulfate and copper.
Heating potassium dichromate with ammonium sulfate
If you mix ammonium sulfate and potassium dichromate and then start heating them, there will also be nitrogen gas in the reaction results of the product components.
![]()
Decomposition of azides
Heating alkali metal azides produces the most chemically pure nitrogen. The process consists in the usual decomposition into simpler components without residue.
![]()